
On this page...
- What is the ASA24 Respondent Nutrition Report (RNR)?
- RNR versions and sample files
- How are ASA24 RNRs generated?
- What are the sources of the nutrition requirements and guidelines used in the ASA24 RNRs?
- Why are respondents asked to provide their sex before their RNR is generated?
What is the ASA24 Respondent Nutrition Report?
The RNR provides feedback to study participants on how their reported intake compares to country-specific dietary guidance for calorie, food group, and nutrient intake. After a respondent completes a recall or record, the researcher can access this report on the ASA24 researcher website and share it with the respondent. During a recall study set-up, researchers can also opt for respondents to receive the RNR immediately after completion. The RNR is available in English and Spanish for US versions, and in English and French for Canadian versions.
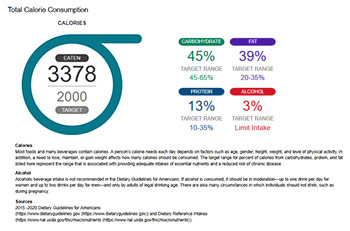
RNR versions and sample files
Version 1
The original RNR was made available in the US version of ASA24-2016 for recalls in January 2017 and for records in August 2017. The RNR was made available in the Canadian version of ASA24-2018 for recalls and records in April 2019.
Version 2
A redesigned RNR was added to recalls and records for the US and Canadian versions of ASA24-2018 and ASA24-2020 (US only) in September 2020. ASA24-Canada-2018 is the most current version of ASA24-Canada.
The redesigned RNR included new text and improved graphics informed by two rounds of user testing to ensure the information provided was useful and well understood. View a PDF [PDF - 2.28 MB] summarizing the findings from the user experience testing. View a Current Developments in Nutrition abstract summarizing the redesigned RNR user testing.
Version 3
A new page was added to the end of the RNR for the US version of ASA24-2022 in November 2023. This page compares nutrient intake from foods and drinks vs. supplements. This addition to the RNR also underwent user testing.
How are ASA24 RNRs generated?
The RNR is available to participants 2 years old and older in studies using the US or Canadian versions of ASA24. To generate an RNR for study participants, the researcher must select the RNR option available on the study details page in the researcher website. Also, respondents must provide their age, sex, and pregnancy and lactation status.
How a respondent views their RNR depends on whether they completed a recall or record:
- If a respondent completed a 24-hour recall, they view their RNR after reporting their intake, before they exit the ASA24 system. Researchers of recall studies can also retrieve the RNR from the researcher site.
- If a respondent completed a food record, the researcher must retrieve the RNR from the researcher website and, if desired, distribute it to the respondent.
Clinicians or researchers using the RNR with populations needing low sodium diets, or anyone interested in how sodium is captured in ASA24, can find more information on sodium measures in the ASA24 system in our Frequently Asked Questions.
What are the sources of the nutrition requirements and guidelines used in the ASA24 RNRs?
The tables and graphs in the RNR compare the respondents’ reported intake to country-specific dietary guidance to give respondents an idea of how their reported intake compares to recommendations for their age and sex group. However, this is an estimate because respondents’ specific food, nutrient, and calorie needs are unique and are influenced by their height, weight, and how much they exercise.
For RNRs generated by the US version of ASA24, the calorie and food group recommendations come from the Dietary Guidelines for Americans and the nutrient requirements from the Institute of Medicine, National Academy of Sciences. The Excel file, “RNR Target Limit Recommendations” provides the recommended daily intake targets and limits for nutrients, food groups, and energy used in the ASA24 Respondent Nutrition Report (RNR). The targets and limits are drawn from the Dietary Reference Intakes (DRI) and Dietary Guidelines for Americans (DGA).
For RNRs generated by the Canadian version of ASA24, the calorie and food group recommendations come from Eating Well with Canada’s Food Guide and nutrient requirements come from the Institute of Medicine, National Academy of Sciences.
Calorie targets in the RNR assume respondents have a moderate level of activity.
Calorie requirements vary widely for pregnant and lactating women based on trimester and other factors. As a result, the estimate for daily calorie goal in this report may not reflect actual calorie needs for pregnant and lactating women. However, the nutrient targets in the RNR reflect the increased requirements for pregnant and lactating women.
Why are respondents asked to provide their sex before their RNR is generated?
The RNR data is based on how the respondent’s dietary intake compares to the recommended intakes for calories, food groups, and specific nutrients from the Dietary Guidelines for Americans and from the Institute of Medicine at the National Academy of Sciences (now the National Academies of Sciences, Engineering, and Medicine). The recommended amounts are based in part on a respondent’s age, sex, and if a respondent is pregnant or lactating.
ASA24 is a registered trademark of HHS.